|
In 1908, Ernest Rutherford, a former student of Thomson's, proved Thomson's raisin bread structure incorrect. These particles were later named electrons.Īfter Eugen Goldstein's 1886 discovery that atoms had positive charges, Thomson imagined that atoms looked like pieces of raisin bread, a structure in which clumps of small, negatively charged electrons (the "raisins") were scattered inside a smear of positive charges. 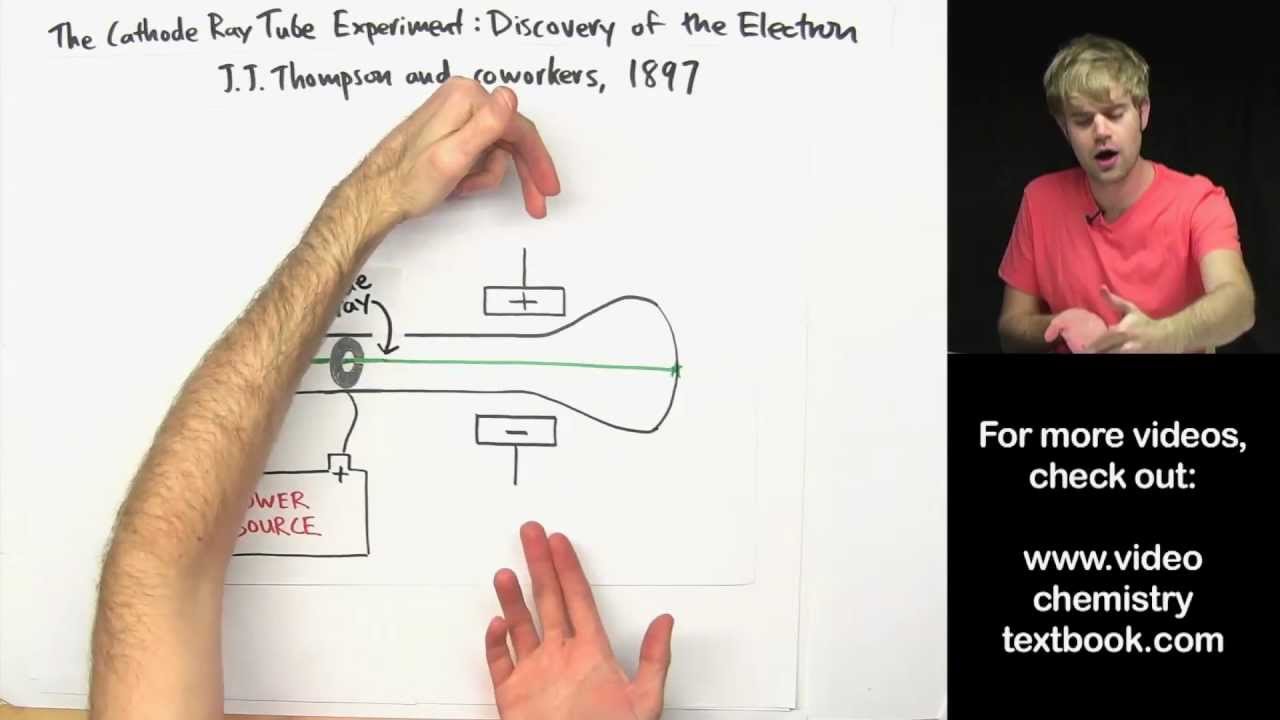
Thomson theorized, and was later proven correct, that the stream was in fact made up of small particles, pieces of atoms that carried a negative charge. Thomson found that the mysterious glowing stream would bend toward a positively charged electric plate. In 1876, Goldstein called them 'cathode rays'. In 1869, Hittorf found that in a very good vacuum (0.01 mm Hg), the Faraday dark space expanded to fill the whole tube, and the cathode emitted rays that caused the glass to glow where they hit. For years scientists had known that if an electric current was passed through a vacuum tube, a stream of glowing material could be seen however, no one could explain why. Cathode Rays The big breakthrough came in the 1850s when Geissler invented a much better (mercury) pump. 5.5K 90K views 4 years ago In 1897, JJ Thomson discovered the electron in his famous cathode ray tube experiment. Thomson during cathode ray tube experiment. A magnetic field of induction 2.4 x 10-3 Wb/m2 is applied at right angles to the electric field and the direction of motion of the electron beam to produce null deflection. It was cathode ray tubes that allowed the English physicist J.J. Cathode rays move opposite to anode rays. Thomson to discover the existence of electrons in 1897.efore we see how J.J. Later it was found that anode rays and Cathode rays both exist at the same time. a negatively charged particle was discovered by J.J. In Thomson’s experiment to find e/m, the deflector plates were 1.2 cm apart and a P.D. JJ Thomson did an experiment with discharge tube using hydrogen gas. For his work on the discovery of electrons he was awarded the Nobel Prize in Physics in 1906. J. Thomson deflected cathode rays with electric and magnetic fields to see the direction they were deflected in and how far they were. 
The air in the hollow glass tube is pumped out to create a vacuum. A cathode ray tube, similar to that used by J. Thomson's notion of the electron came from his work with a nineteenth century scientific curiosity: the cathode ray tube. Thomson was a British physicist, and was born in Cheetham Hill, a suburb of Manchester, on 18 December 1856. In 1897, age 40, Thomson carried out a now famous experiment with a cathode ray tube. Thomson's work suggested that the atom was not an "indivisible" particle as John Dalton had suggested but a jigsaw puzzle made of smaller pieces. Thomson dramatically changed the modern view of the atom with his discovery of the electron.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
